MTS Spark Waves® – a Novel Therapy for Coronary Heart Diseases (CHD)
Epidemiology
Cardiovascular disease (CVD) is the leading cause of deaths and disability-adjusted life years globally. Coronary artery disease (CAD) -also called ischemic heart disease (IHD)- is the most prevalent CVD which affected 110 million people and resulted in 8.9 million deaths in 2015. It makes up 15.6 % of all deaths, making it the most common cause of death globally. 1,2
Definition and Pathophysiology:
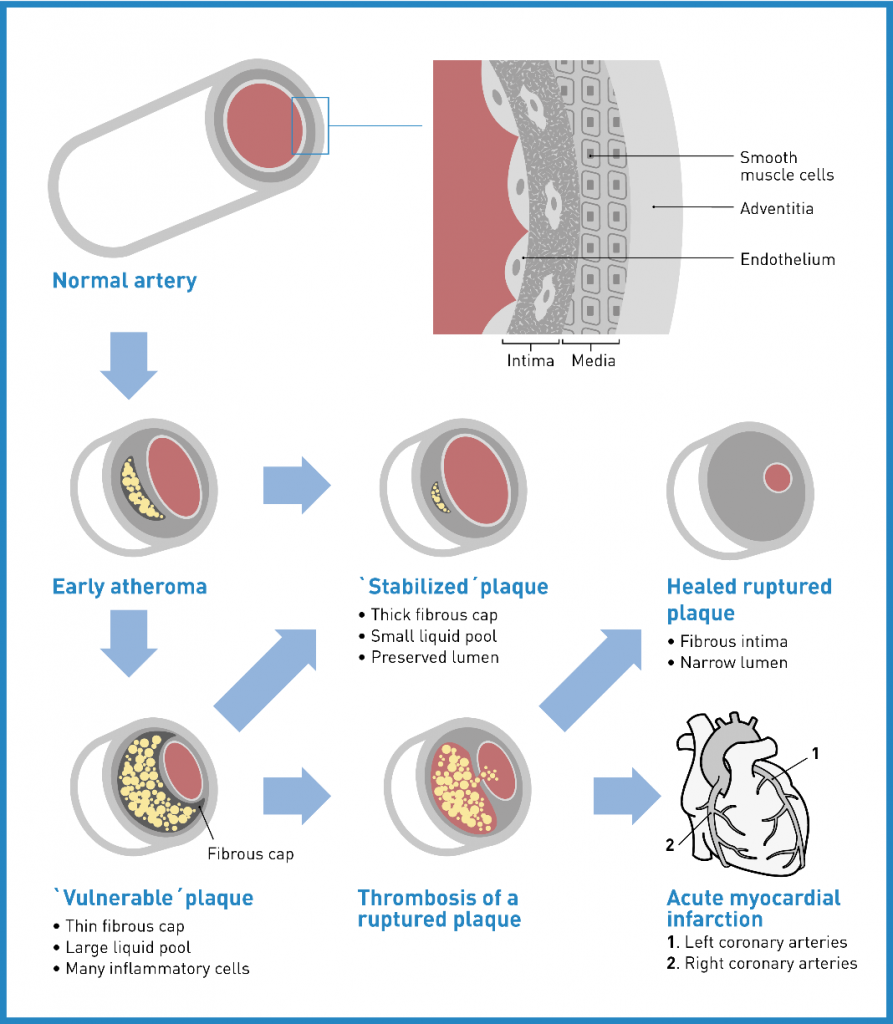
IHC is characterised by an impaired blood -and therefore oxygen and nutrient- supply to the heart (= myocardial ischemia). The most common cause of this impairment is vascular injury due to cholesterol plaques in the arteries, known as atherosclerosis. Affected persons experience a variety of symptoms like chest pain (angina pectoris, most common symptom), heaviness, tightness, burning, pain in the arms or shoulders, shortness of breath or sweating. Atherosclerotic plaque rupture with thrombosis can lead to complete restriction of blood flow with subsequent tissue necrosis and is the pathologic mechanism responsible for acute myocardial infarctions (AMI) and sudden coronary death (SCD). The main risk factors for development of CAD are hypertension, cigarette smoking, excessive alcohol consumption, diabetes mellitus (or elevated glucose levels), elevated cholesterol levels, lack of exercise, being overweight and emotional stress. 3,4
Diagnosis and Management
Common tests to be performed for diagnostic purposes include electrocardiogramm, echocardiogramm, cardiac stress testing, coronary computed tomographic angiography and coronary angiogramm.
Self-care strategies are used to guide patients in order to reduce risks and to stabilize or prevent symptoms. This comprises medication adherence, symptom monitoring, dietary adherence, fluid restriction, alcohol restriction, weight monitoring, smoking cessation and exercise. 5
Medications for diabetes, high cholesterol, or high blood pressure are administered. Additional medications such as antiplatelets (e. g. aspirin), beta blockers or nitroglycerin may be recommended. Besides optimized drug therapy and lifestyle changes, revascularization is the gold standard after AMI. At an advanced stage, procedures such as percutanous coronary intervention (PCI, e. g. balloon angioplasty or stents) or coronary artery bypass grafting (CABG) are performed in order to re-establish blood supply of the myocardial tissue, though still limited by non-viable myocardium. Especially in the case of the steadily increasing number of patients that are suffering from refractory angina (RFA) -defined as „a persistent painful condition which cannot be controlled by a combination of medication, PCI or GABG“- the prognosis remains poor, due to the lack of effective treatments. Several new alternative treatment methods are being investigated; a number of studies showed that transmyocardial and percutaneous myocardial laser revascularization TMLR), spinal cord stimulation and stem cell therapy may reduce angina symptoms and improve exercise capacity. Nevertheless, these treatment modalities are invasive, cost expensive or still at a preclinical stage. Transmyocardial laser revascularization (TMLR), the most studied alternative therapy, is associated with significant early postoperative mortality risk. 6,7
It is therefore crucial to develop effective therapeutic strategies and the medical community is urgently looking for innovative ways to approach angiogenesis and regeneration of the ischemic / infarcted heart tissue!
Cardiac shock wave therapy (CSWT)
is a promising non-invasive modality for patients with stable CAD and has been successfully applied in cardiovascular medicine since 1999 in a considerable number of clinical studies.
Spark Wave® Therapy – Mode of Action in Coronary Heart Diseases
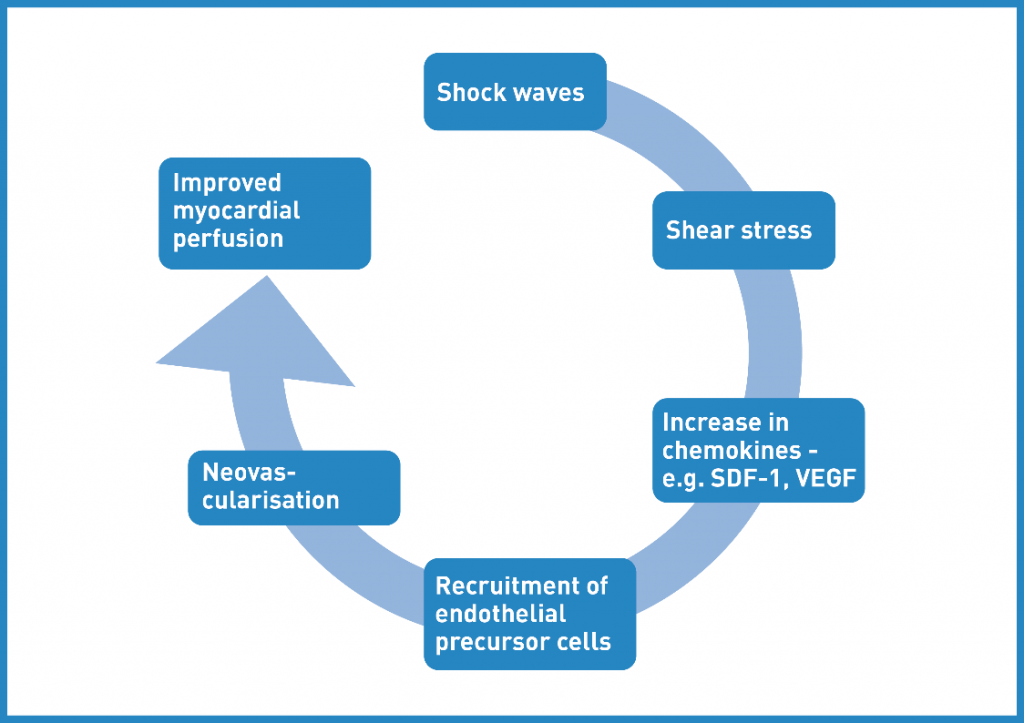
At low energy levels, SWT is routinely used for the treatment of soft tissue injuries and non-healing bone fractures. The regenerative effects of SWT have been mainly attributed to the mediation and stimulation of regeneration-associated processes by induction of growth factor expression. Mechanotransduction -the translation of the physical stimulus to a biological response- is assumed to be the underlying mechanism. Most importantly, SWT leads to angiogenesis and neovascularization. Therefore, extensive research has been performed on cardiac SWT. 8
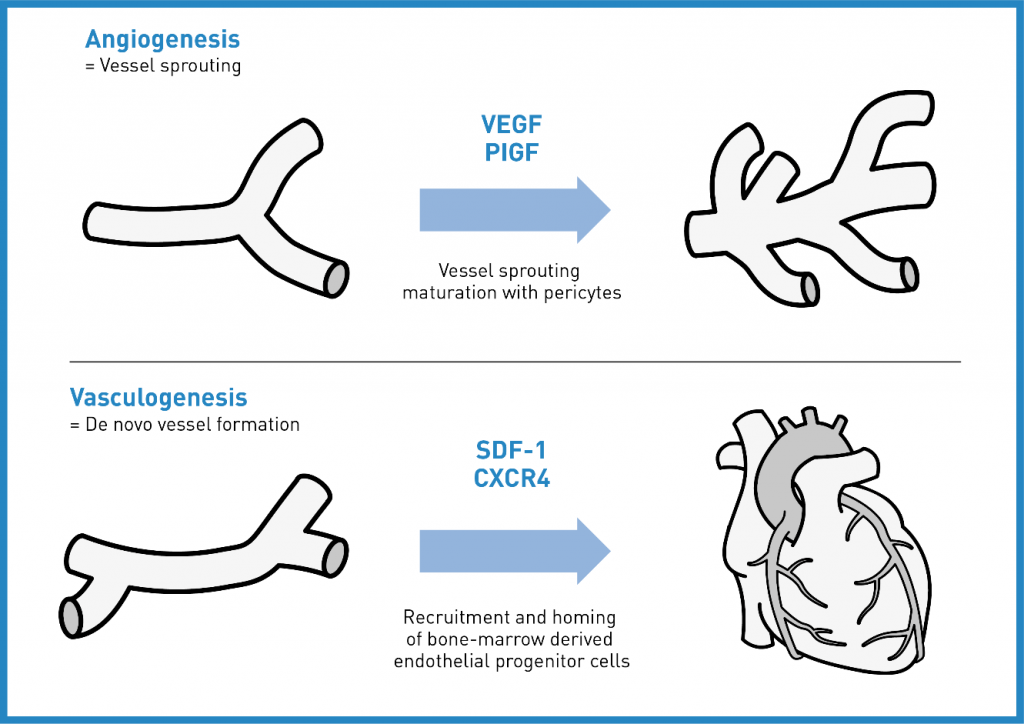
Angiogenesis -the sprouting of new capillaries from pre-existing blood vessels- starts with the release of vascular endothelial growth factors (VEGFs) which control the formation of new blood vessels. Maturation of vessels from capillaries to arterioles is mainly triggered by the placental growth factor (PLGF).9
Vasculogenesis -the de novo development of vessels- is performed by endothelial progenitor cells (EPCs) and vascular progenitor cells. The most important chemoattractant for the mobilization and recruitment of EPCs is stromal cell derived factor-1 (SDF-1).10
SWT stimulates both. Vasculogenesis is the growth of new blood vesssels and angiogenesis is responsible for the remodelling and expansion of this network.
Clinical Status of Cardiac Shock Wave Therapy
CSTW in the animal model (rat, mouse, pig) indicated angiogenesis, i. e. more vital cells, recruitment of endothelial progenitor cells, higher number of vessels and increased microvascular density. The upregulation of pro-angiogenic growth factors, e. g. VEGF, PLGF and SDF-1, was demonstrated. Angiogenesis as well as wound healing depend on inflammation in a specific timely manner. Moreover, Angiogenesis and inflammation are capable of influencing and potentiating each other. The clue of CSWT is modulation of inflammation in at least a bi-phasic manner over time in order to create the milieu for regeneration. 11,12
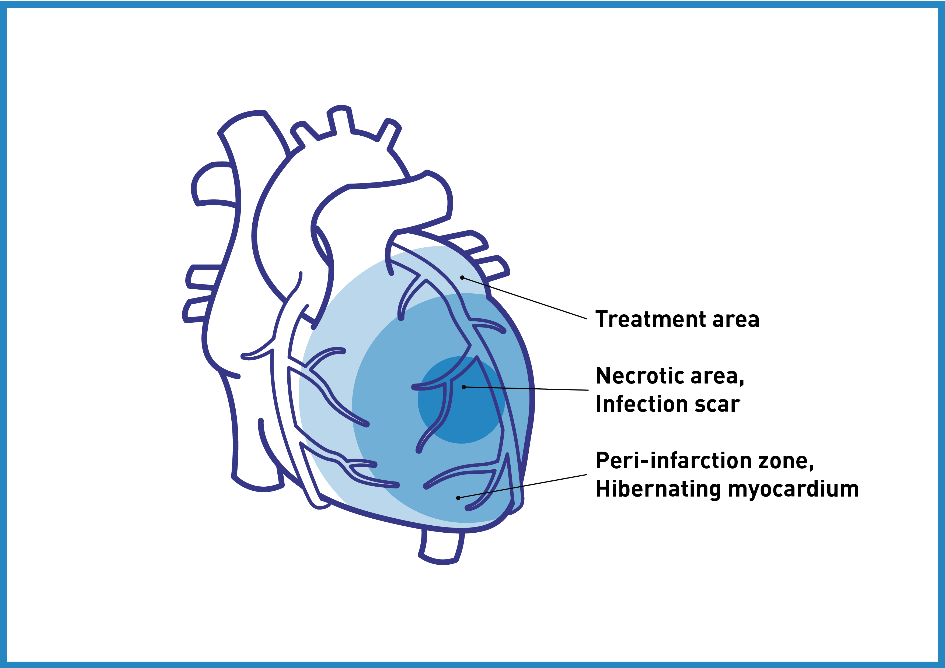
CSWT in the clinical context is performed using a SW device coupled with a cardiac ultrasound targeting system and triggered under electro-cardiographic R-Wave gating. For optimal therapy, the treatment area is divided into target zones corresponding to the focal zone size of the SW applicator. 6
Direct epicardial shock wave therapy with MTS technology has been utilized in the course of coronary artery bypass graft surgery (CABG) in a clinical pilot trial:
https://clinicaltrials.gov/ct2/show/NCT00741065
Currently, another prospective, single-blind, randomized, single-centre study is being conducted at the Clinical Trial Center of the University of Innsbruck to investigate the safety and performance characteristics of the Direct Epicardial Shockwave Therapy System (DESWT) as applied with the Cardiac Shockwave Probe (CSP).
https://clinicaltrials.gov/ct2/show/NCT03859466
This technique bypasses the potential risk of lung injury but the surgical access is still very invasive and direct epicardial SWT can therefore be used for CABG patients only. 13
To date, several RCTs have demonstrated promising clinical outcomes without serious adverse effects. Latest systematic review and meta-analysis of CSWT studies in stable CAD demonstrated consistent improvement of clinical variables:6
-
- Significant improvement of left ventricular (LV) function
- Significant improvement of myocardial perfusion
- Significant increase of angiogenesis markers
perfusion. SDF-1 = stromal cell-derived factor; VEGF = vascular endothelial growth factor. (Taken based on 15 )
SWT represents a promising regenerative treatment option for patients with ischemic cardiomyopathy. It could serve as a promotor of „endogenous cell therapy“, without exhibiting the disadvantages of cell harvesting necessary for conventional cell therapy. It is non-invasive, cost-effective and has no side effects. CSWT could be translated efficiently into the clinical setting and is a promising option even for patients with end-stage CAD. Multicenter adequately powered randomized double blind studies are warranted.6,8
Clinical Implication – Cardiac Shock Wave Therapy for Coronary Heart Diseases:
- Improvement of angina symptoms
- Improvement of heart function
- Improvement of myocardial perfusion
- Improvement of exercise capacity
- Improvement of quality of life
- No complications or adverse effects were observed
Benefits of MTS Spark Wave® Therapy at a Glance:
- Activation of endogenous growth factors (e. g. VEGFs, PLGF, BNDF, BMP, FGF-2, IGF-1, NO, PCNA, TGF-ß)
- Induction of stem cell proliferation and migration
- Stimulation of neovascularisation and angiogenesis
- Enhancement of blood circulation and cell metabolism
- Reduction of cell apoptosis and tissue necrosis
- Modulation of the inflammatory response
- Stimulation of the catabolism and natural anabolic growth function of tissues (skin, bones, tendons, joints, muscles, nerves and many more)
- Promoting tissue remodelling and regeneration
- Amelioration of myocardial fibrosis
→ CSWT as an effective, safe and non-invasive approach to treat refractory angina?!
Spark Wave® Therapy References for Cardiology:
Gollmann-Tepeköylü, C. et al. miR-19a-3p containing exosomes improve function of ischaemic myocardium upon shock wave therapy.Cardiovasc. Res. (2019). doi:10.1093/cvr/cvz209
| Title | Author | Journal | Year of Publication |
| Shock waves induce angiogenesis via exosome release | Graber et al. | 20th ISMST Congress in San Sebastian, Spain
Abstract P25 |
2017 |
| Elucidating the molecular mechanisms underlying cardiac shock wave therapy | Szwarc et al. | 20th ISMST Congress in San Sebastian, Spain
Abstract P28 |
2017 |
| The beneficial effects of in vitro shock wave treatment on cardiomyogenesis are energy dependent | Fuchs et al. | 4th ISMST Basic Research Meeting in Vienna, Austria | 2016 |
| Shock wave therapy causes increased macrophage recruitment and enhances M2 polarization | Nägele et al. | 4th ISMST Basic Research Meeting in Vienna, Austria | 2016 |
References
- Wang, H. et al. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet (2016). doi:10.1016/S0140-6736(16)31012-1
- Joseph, P. et al. Reducing the global burden of cardiovascular disease, part 1: The epidemiology and risk factors. Circulation Research (2017). doi:10.1161/CIRCRESAHA.117.308903
- Wong, N. D. Epidemiological studies of CHD and the evolution of preventive cardiology. Nature Reviews Cardiology (2014). doi:10.1038/nrcardio.2014.26
- Mendis, Shanthi; Puska, Pekka; Norrving, B. Global atlas on cardiovascular disease prevention and control (1st ed.). Geneva World Heal. Organ. Collab. with World Hear. Fed. World Stroke Organ. 3–18 (2011).
- Jackevicius CA, Page RL, Buckley LF, Jennings DL, Nappi JM, S. A. Key Articles and Guidelines in the Management of Heart Failure: 2018 Update. J Pharm Pr. 32, 77–92 (2019).
- Burneikaitė, G. et al. Cardiac shock-wave therapy in the treatment of coronary artery disease: systematic review and meta-analysis. Cardiovasc. Ultrasound (2017). doi:10.1186/s12947-017-0102-y
- Li H, L. M. Cardiac shock wave therapy: an alternative non-invasive therapy for refractory angina. Eur Rev Med Pharmacol Sci 22, 5402–5410 (2018).
- Gollmann-Tepeköylü, C. et al. Shock wave therapy improves cardiac function in a model of chronic ischemic heart failure: Evidence for a mechanism involving vegf signaling and the extracellular matrix. J. Am. Heart Assoc. (2018). doi:10.1161/JAHA.118.010025
- Carmeliet, P. Mechanisms of angiogenesis and arteriogenesis. Nature Medicine (2000). doi:10.1038/74651
- Dimmeler, S. ATVB in focus: Novel mediators and mechanisms in angiogenesis and vasculogenesis. Arteriosclerosis, Thrombosis, and Vascular Biology (2005). doi:10.1161/01.ATV.0000187471.06942.17
- Holfeld, J., Lobenwein, D., Tepek??yl??, C. & Grimm, M. Shockwave therapy of the heart. International Journal of Surgery 24, 218–222 (2015).
- Holfeld, J. et al. Shockwave therapy differentially stimulates endothelial cells: Implications on the control of inflammation via toll-like receptor 3. Inflammation 37, 65–70 (2014).
- Zimpfer, D. et al. Direct epicardial shock wave therapy improves ventricular function and induces angiogenesis in ischemic heart failure. J. Thorac. Cardiovasc. Surg. 137, 963–70 (2009).
- Libby, P. Inflammation in atherosclerosis. Nature (2002). doi:10.1038/nature01323
- 15. Cheng, K. & Silva, R. de. New advances in the management of refractory angina pectoris. Eur. Cardiol. Rev. (2018). doi:10.15420/ecr.2018:1:2